
The aim of this essay is to prove if there equivalence between these two statements and it will be achieved by analysing them. It is the second law of thermodynamics which provides the criterion as to the probability of various processes through the statements of Clausius that ‘’Heat does not pass from a body at low temperature to one at high temperature without an accompanying change elsewhere’’ and Kelvin that ‘’No cyclic process is possible in which heat is taken from a hot source and converted completely into work’’. 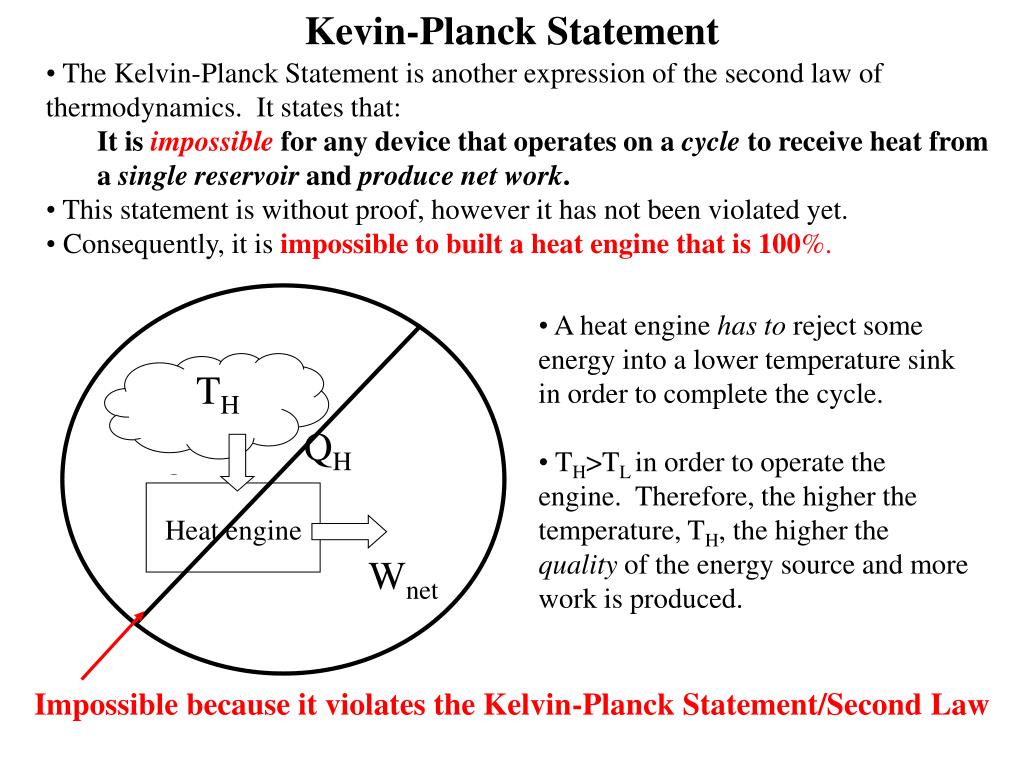
But it does not indicate whether that change of state or the process is at all feasible or not. The first law of thermodynamics states that a certain energy balance will hold when a system undergoes a change of state or a thermodynamic process.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
